Aldehydes And Ketones Formula
Aldehydes And Ketones Formula. Make the most out of the aldehyde and ketone cheat sheet over here and get good grip on the concept. The carbon atom exhibits sp2 hybridization.
 PPT ALDEHYDES AND KETONES PowerPoint Presentation ID from www.slideserve.com
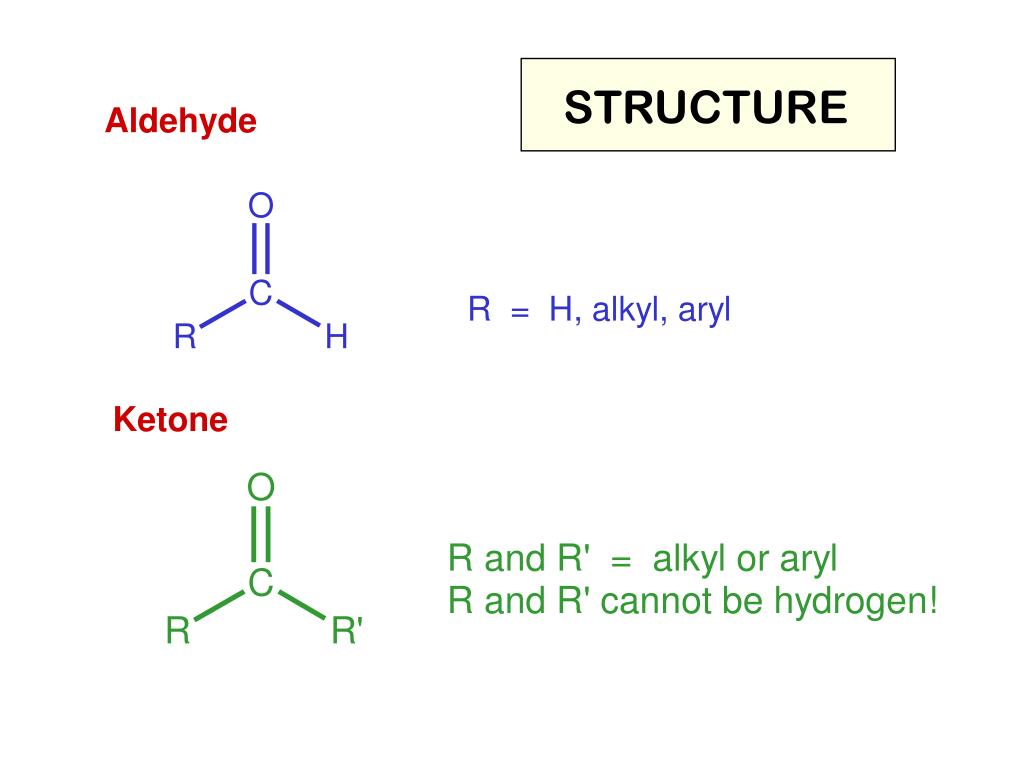
PPT ALDEHYDES AND KETONES PowerPoint Presentation ID from www.slideserve.com(i) for both of these. An aldehyde group always lies at the end of a carbon chain. The general molecular formula of aldehydes and ketones is:

The general formula of alkene is c n h2 n+1 so the general formula for aldehyde will be c n h2 n+1 cho or c n h 2n o. Aldehydes and ketones are both carbonyl compounds, that is they contain alkyl chains attached to a c=o group.

Where r may be an alkyl, alkenyl, alkynyl or aryl group. Make the most out of the aldehyde and ketone cheat sheet over here and get good grip on the concept.

The result is the formation of a bond between the carbon of one mole of carbonyl compound and the carbonyl carbonyl of a second, to give a hydroxy aldehyde or ketone. In aldehydes, the carbonyl group has one hydrogen atom attached to it together with either a 2nd hydrogen atom or a hydrogen group which may be an alkyl group or one containing a benzene ring.
![Reduction of Aldehydes and Ketones [PPT Powerpoint]](https://static.documents.pub/img/1200x630/reader012/image/20180902/56814944550346895db68e48.png?t=1606757912)
The aldehyde shares a carbonyl group (c=o) with several other organic functional groups like ketones, carboxylic acids, and esters. Where r may be an alkyl, alkenyl, alkynyl or aryl group.
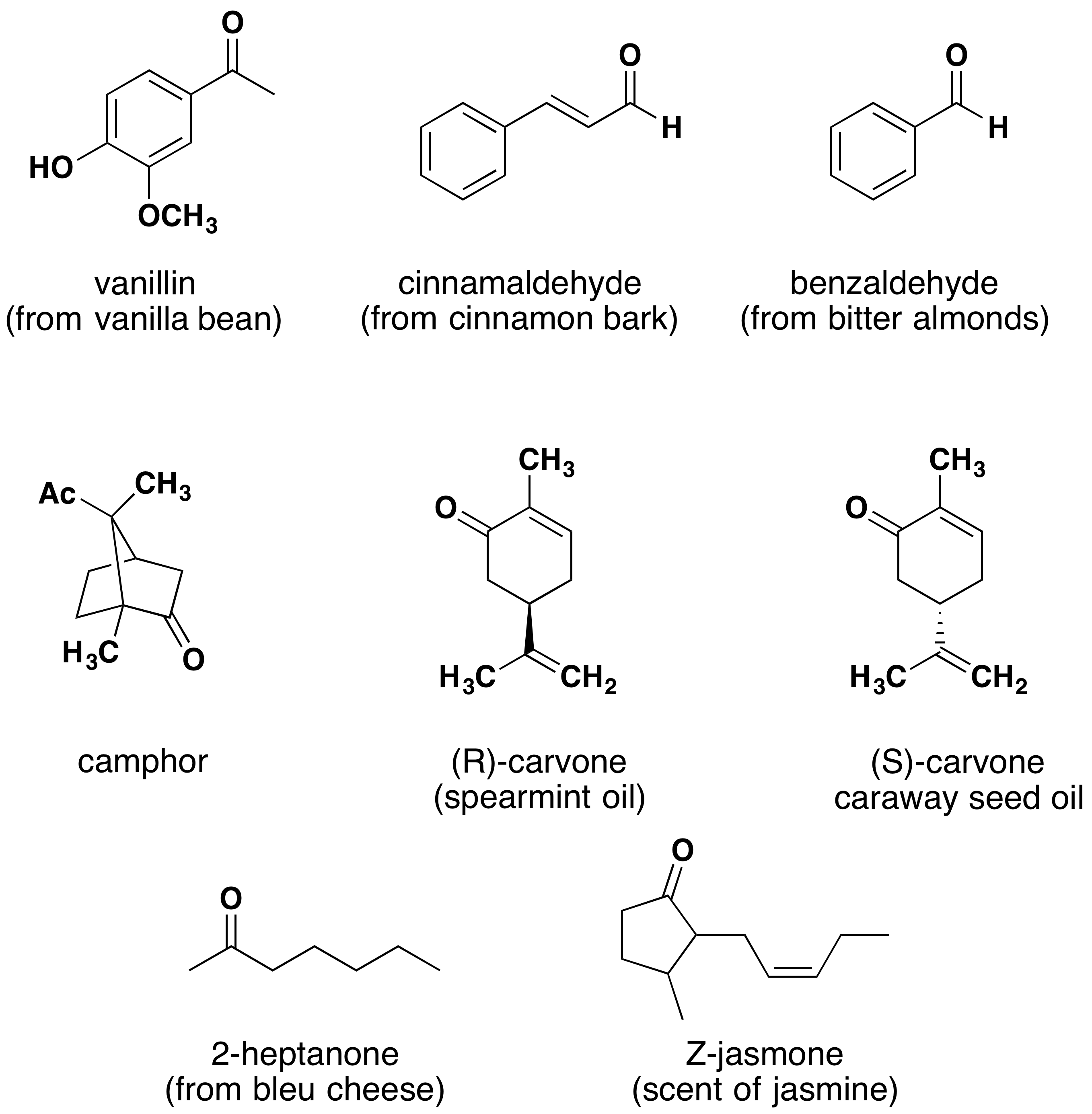
For 7 & 8 g.m.p. The general formula of alkene is c n h2 n+1 so the general formula for aldehyde will be c n h2 n+1 cho or c n h 2n o.

Aldehydes and ketones with more than six carbons are essentially insoluble in water. Aldehyde and ketones both consist of the carbonyl group ( > c = o).

Aid and ketone ☒ for 7 & 8 g.m.p. The total number of aldehydes and ketones with the molecular formula c4h8o is.
![ALDEHYDES AND KETONES [PPT Powerpoint]](https://reader020.fdocuments.in/reader020/slide/20190918/56813354550346895d9a654d/document-11.png?t=1603987063)
Aldehyde has the structure rch(=o) while a ketone has the structure of r 2 c(=o). The total number of aldehydes having the molecular formula c 5 h 1 0 o is :

Aldehydes and ketones sct page 8 of 13 mark schemes q1. The reaction is catalyzed by acids and bases.
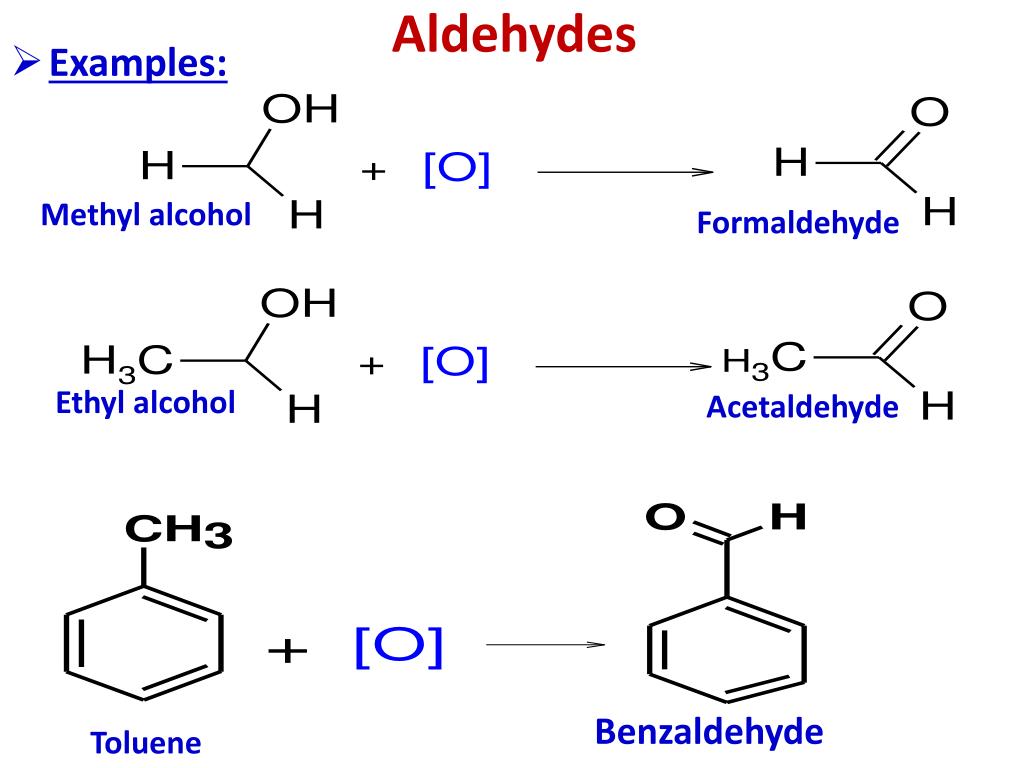
This gives aldehydes the general formula. In both aldehydes and ketones, the geometry around the carbon atom in the carbonyl group is trigonal planar;
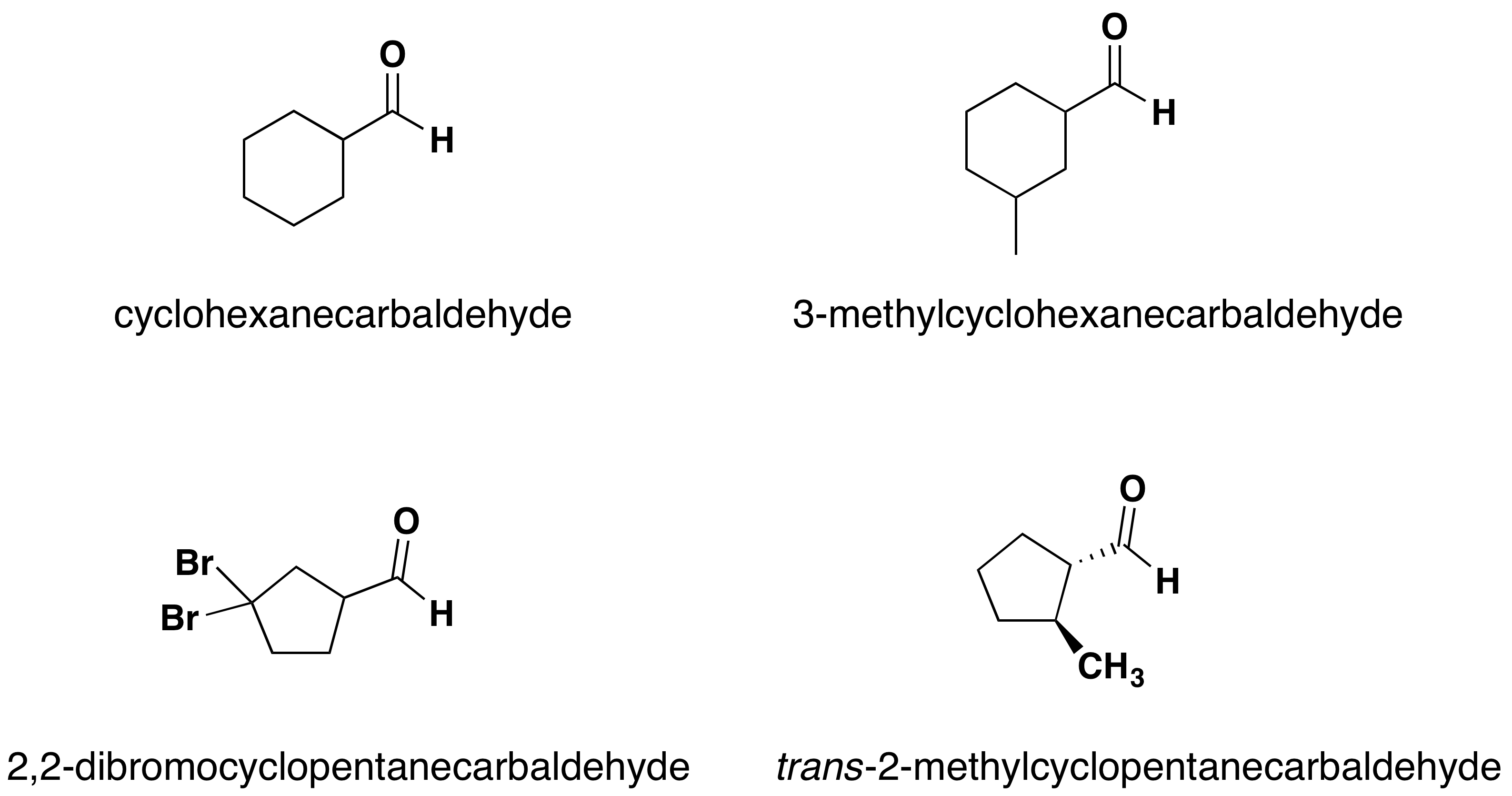
This gives aldehydes the general formula. Other aldehydes in ketones in the solution.

Make the most out of the aldehyde and ketone cheat sheet over here and get good grip on the concept. Bandict reagent (cuso 4 + naoh + sod.citrate) → red ppt.
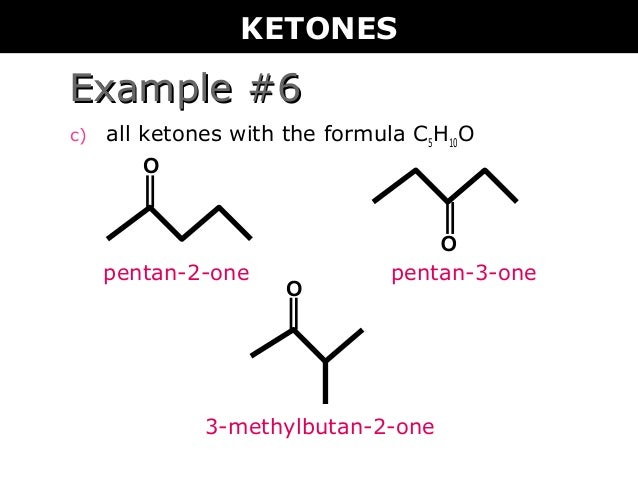
Aid and ketone ☒ for 7 & 8 g.m.p. (i) for both of these gmp:

Adding water to an aldehyde or ketone produces a product called a hydrate or gemdiol (two oh groups on one carbon). The general formula of alkene is c n h2 n+1 so the general formula for aldehyde will be c n h2 n+1 cho or c n h 2n o.

The reaction is catalyzed by acids and bases. The total number of aldehydes having the molecular formula c 5 h 1 0 o is :

Legal test → methyl ketone hcho☒ ch 3 cho with alk. The carbon atom exhibits sp2 hybridization.

The total number of aldehydes and ketones with the molecular formula c4h8o is. Legal test → methyl ketone.
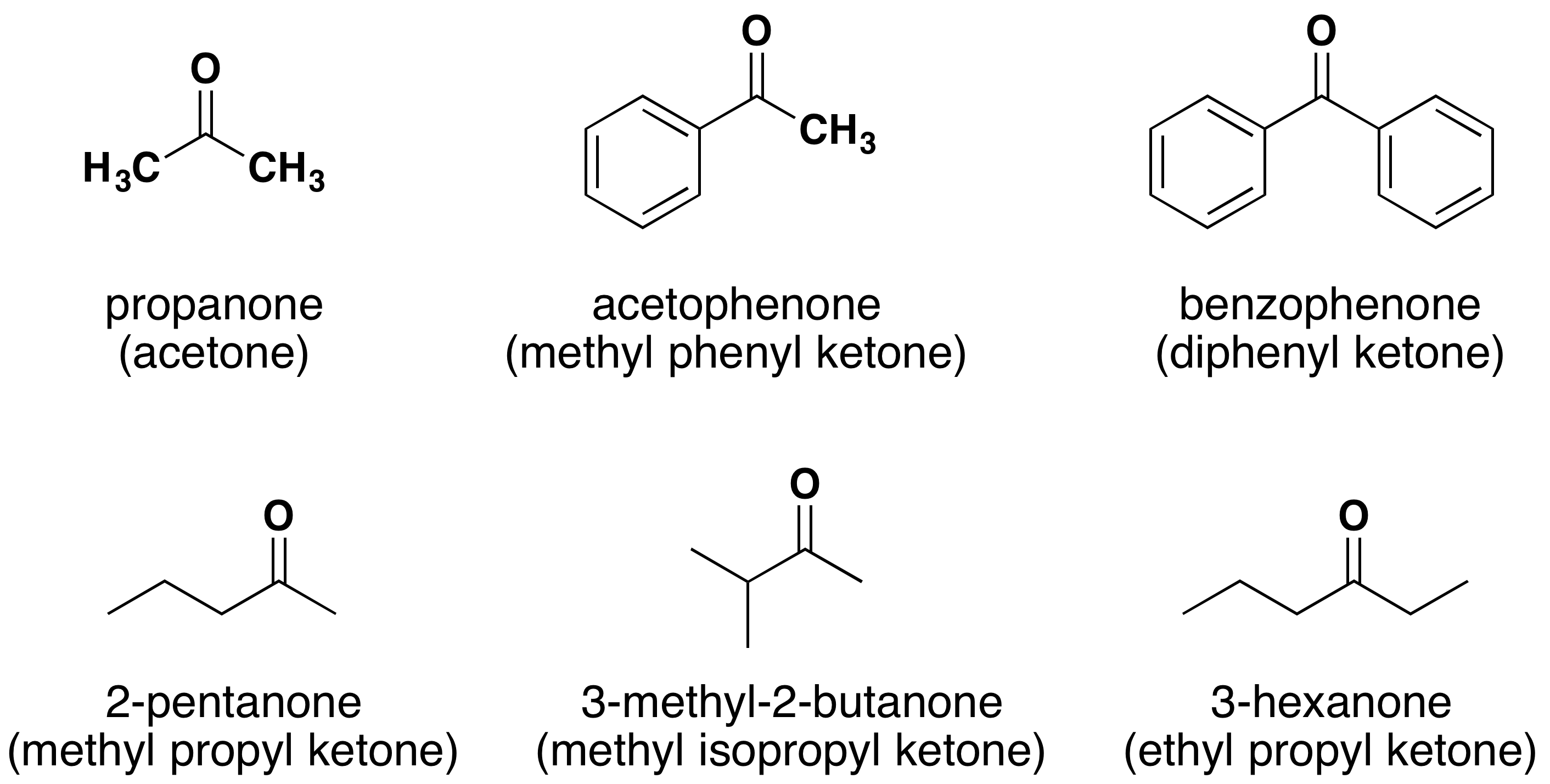
Where r may be an alkyl, alkenyl, alkynyl or aryl group. Learn more about the definition of aldehyde, its formula, chemical structure, how it.
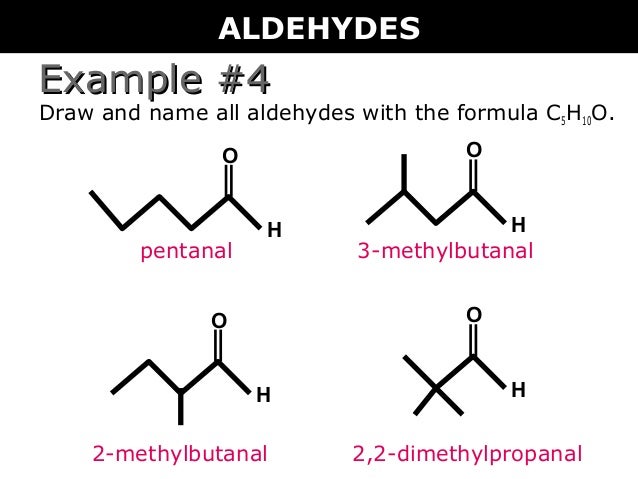
Legal test → methyl ketone hcho☒ ch 3 cho with alk. Nitroprusside → red violet colour.

Aid and ketone ☒ for 7 & 8 g.m.p. Bandict reagent (cuso 4 + naoh + sod.citrate) → red ppt.
The Result Is The Formation Of A Bond Between The Carbon Of One Mole Of Carbonyl Compound And The Carbonyl Carbonyl Of A Second, To Give A Hydroxy Aldehyde Or Ketone.Aldehyde and ketones both consist of the carbonyl group ( > c = o). The physical properties of these carbonyl compounds primarily depend on the nature of the carbonyl group. (i) for both of these gmp:
The Carbon Atom Exhibits Sp2 Hybridization.Legal test → methyl ketone. Aldehydes and ketones are both carbonyl compounds, that is they contain alkyl chains attached to a c=o group. Aid and ketone ☒ for 7 & 8 g.m.p.
The Total Number Of Aldehydes Having The Molecular Formula C 5 H 1 0 O Is :The general molecular formula of the homologous aldehyde series is \({{\rm{c}}_{\rm{n}}}{{\rm{h}}_{{\rm{2n + 1}}}}{\rm{cho,}}\) where \({\rm{n = 1,2,3}}……\) Aldehydes and ketones sct page 8 of 13 mark schemes q1. (b) give structures and names (common or iupac substitutive names) for all the aldehydes and ketones that contain a benzene ring.
The Lower Aldehydes And Ketones Are Soluble In Water.These may be the same or different. This gives aldehydes the general formula. The general formula of alkene is c n h2 n+1 so the general formula for aldehyde will be c n h2 n+1 cho or c n h 2n o.
This Condensation Reaction Between Two Moles Of An Aldehyde Or Ketone Is Called The Aldol Condensation.For 7 & 8 g.m.p. In aldehydes, the carbonyl group has one hydrogen atom attached to it together with either a 2nd hydrogen atom or a hydrogen group which may be an alkyl group or one containing a benzene ring. Where r may be an alkyl, alkenyl, alkynyl or aryl group.
Belum ada Komentar untuk "Aldehydes And Ketones Formula"
Posting Komentar